Research Article |
|
Corresponding author: Quan Lu ( luquan@caf.ac.cn ) Corresponding author: HuiXiang Liu ( hxliu@sdau.edu.cn ) Academic editor: Marc Stadler
© 2018 HuiMin Wang, YingYing Lun, Quan Lu, HuiXiang Liu, Cony Decock, XingYao Zhang.
This is an open access article distributed under the terms of the Creative Commons Attribution License (CC BY 4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation:
Wang HM, Lun YY, Lu Q, Liu HX, Decock C, Zhang XY (2018) Ophiostomatoid fungi associated with pines infected by Bursaphelenchus xylophilus and Monochamus alternatus in China, including three new species. MycoKeys 39: 1-27. https://doi.org/10.3897/mycokeys.39.27014
|
Abstract
The activity of the pine wood nematode Bursaphelenchus xylophilus leads to extremely serious economic, ecological and social losses in East Asia. The nematode causes pine wilt disease, which is currently regarded as the most important forest disease in China. The pathogenic nematode feeds on dendrocola fungi to complete its cycle of infection. As the vector of the nematode, the Japanese pine sawyer (Monochamus alternatus) also carries dendrocola fungi. Pine woods, infected by B. xylophilus and tunnelled by M. alternatus, are also inhabited by ophiostomatoid fungi. These fungi are well known for their association with many bark and ambrosia beetles. They can cause sapstain and other serious tree diseases. The aims of our study were to investigate and identify the ophiostomatoid communities associated with the epidemic pine wood nematode and the pine sawyer in Pinus massoniana and P. thunbergii forests, which are the main hosts of the pine wood nematode in China. Two hundred and forty strains of ophiostomatoid fungi were isolated from nematode and sawyer–infected trees in the coastal Shandong and Zhejiang Provinces, representing newly and historically infected areas, respectively. Six ophiostomatoid species were identified on the basis of morphological, physiological and molecular data. For the latter, DNA sequences of the internal transcribed spacer (ITS1–5.8S–ITS2) region and partial b-tubulin gene were examined. The ophiostomatoid species included one known species, Ophiostoma ips, three novel species, viz. Ophiostoma album sp. nov., Ophiostoma massoniana sp. nov. and Sporothrix zhejiangensis sp. nov. and two species whose identities are still uncertain, Ophiostoma cf. deltoideosporum and Graphilbum cf. rectangulosporium, due to the paucity of the materials obtained. The ophiostomatoid community was dominated by O. ips. This study revealed that a relatively high species diversity of ophiostomatoid fungi are associated with pine infected by B. xylophilus and M. alternatus in China.
Keywords
Ophiostoma , taxonomy, Sporothrix , Ophiostoma minus complex, Ophiostoma ips complex
Introduction
The pathogenic pine wood nematode (PWN) Bursaphelenchus xylophilus (Steiner & Buhrer) Nickle (Aphelenchida, Parasitaphelenchidae), presumably native to North America (
Bursaphelenchus xylophilus infects many species of coniferous trees, mainly from the genus Pinus (
The ophiostomatoid fungi are one of the most common fungal groups inhabiting wood infected by B. xylophilus. Further, many ophiostomatoid reproduction structures are detected in the tunnels of M. alternatus, suggesting a relationship between the fungi and the occurrence and development of the disease. For instance, O. ips has been found in the PWN vector beetles in North America, China and Korea (
The association of PWN with ophiostomatoid fungi and bacteria likely contributes to the nematode’s pathogenicity (
Thus far, the association with PWN and Monochamus spp. has been documented for only five species of ophiostomatoid fungi worldwide (
The aims of the current study were to investigate and identify the ophiostomatoid mycobiota associated with the nematode and sawyer in the epidemic forests of Shandong and Zhejiang Provinces in eastern China to facilitate the understanding of pine wilt disease infection and prevalence mechanisms. The two coastal provinces, Shandong and Zhejiang, represent new and historic epidemic areas, with P. thunbergii and P. massoniana as hosts, respectively.
Materials and methods
Collection of samples and fungus isolations
Fungi were isolated from 98 samples of M. alternatus galleries or pupal chambers in P. massoniana and P. thunbergii in the Zhejiang and Shandong Provinces (Table
Strains of ophiostomatoid fungi isolated from pines infested by Monochamus alternatus and pine wood nematode in the current study.
| Group | Species | Strain No. | Host | Origin (Latitude, Longitude) | Genbank No. | Collector | |
|---|---|---|---|---|---|---|---|
| ITS | β-tubulin | ||||||
| A | Sporothrix zhejiangensis sp. nov. | MUCL 55181 (CFCC52167, CXY1612) | Pinus massoniana | Yuyao, Zhejiang (29°58'10.2"N, 121°05'57.1"E) | KY094069 | MH397728 | Q. Lu, YY Lun |
| MUCL 55182 (CFCC52164, CXY1613) | P. massoniana | Yuyao, Zhejiang (29°58'10.2"N, 121°05'57.1"E) | KY094070 | MH397729 | |||
| MUCL 55183 (CFCC52165, CXY1614) | P. massoniana | Yuyao, Zhejiang (29°58'10.2"N, 121°05'57.1"E) | KY094071 | MH397730 | |||
| MUCL 55184 (CFCC52166, CXY1615) | P. massoniana | Yuyao, Zhejiang (29°58'10.2"N, 121°05'57.1"E) | KY094072 | MH397731 | |||
| B | Ophiostoma album sp. nov. | MUCL 55189 (CFCC52168, CXY1622) | P. massoniana | Yuyao, Zhejiang (29°58'10.2"N, 121°05'57.1"E) | KY094073 | MH360979 | |
| MUCL 55190 (CFCC52169, CXY1642) | P. massoniana | Yuyao, Zhejiang (29°58'10.2"N, 121°05'57.1"E) | KY094074 | MH360980 | |||
| CFCC52170 (CXY1643) | P. massoniana | Yuyao, Zhejiang (29°58'10.2"N, 121°05'57.1"E) | KY094075 | MH360981 | |||
| C | Ophiostoma ips | CXY1628 | P. thunbergii | Changdao, Shandong (37°59'13.5"N, 120°42'18.1"E) | KY593324 | MH324804 | |
| CXY1631 | P. thunbergii | Zhoushan, Zhejiang (29°52'51.33"N, 122°24'14.13"E) | MH324811 | MH324805 | |||
| CXY1635 | P. massoniana | Yuyao, Zhejiang (29°58'10.2"N, 121°05'57.1"E) | MH324812 | MH324808 | |||
| CXY1638 | P. thunbergii | Fuyang, Zhejiang (30°05'15.1"N, 119°58'55.1"E) | MH324813 | MH324809 | |||
| CXY1639 | P. massoniana | Weihai, Shandong (37°23'23.6"N, 122°32'33.1"E) | MH324814 | MH324810 | |||
| D | Ophiostoma massoniana sp. nov. | MUCL 55179 (CFCC51648, CXY1610) | P. massoniana | Fuyang, Zhejiang (30°05'15.1"N, 119°58'55.1"E) | KY094067 | MH370810 | |
| MUCL 55180 (CFCC51649, CXY1611) | P. massoniana | Yuyao, Zhejiang (29°59'36.87"N, 121°09'09.90"E) | KY094068 | MH370811 | |||
| E | Graphilbum cf. rectangulosporium | CXY1623 | P. massoniana | Yuyao, Zhejiang (29°59'36.87"N, 121°09'09.90"E) | MH324816 | – | |
| F | Ophiostoma cf. deltoideosporum | MUCL 55191 (CXY1640) | P. thunbergii | Weihai, Shandong (37°23'23.6"N, 122°32'33.1"E) | MH324815 | – | |
Culture and morphological studies
The ophiostomatoid fungal strains were incubated on 2% MEA and 2% potato dextrose agar (PDA; 200 g potato and 20 g dextrose, 20 g agar powder in 1000 ml of deionised water: the dextrose was obtained from American Amresco) in the dark at 25 °C in an incubator. Fungal growth on MEA plates was monitored daily. Hyphal tips of emerging colonies were transferred to fresh MEA plates to purify the fungi. Slides were made to observe the sexual/asexual state structures; these were mounted in lactic acid cotton blue on glass slides and examined under a BX51 OLYMPUS microscope. Fifty measurements were made of each microscopic taxonomically informative structure. The measurements are presented in the form: (minimum–) mean minus standard deviation–mean plus standard deviation (–maximum).
A 5-mm mycelium disc was cut from an actively growing fungal colony using a sterile cork borer and placed at the centre of MEA plates, with the aerial mycelium side in contact with the medium. Three replicate plates were prepared for each strain and were incubated at temperatures ranging from 5–40 °C at five-degree intervals. The colony diameters on each Petri dish were determined along two perpendicular axes every day until the entire dish was covered. The colour descriptions were provided according to
DNA extraction, PCR and sequencing reactions
DNA was extracted from freshly collected mycelia grown in liquid malt medium (20g malt extract in 1000 ml of deionised water) at 25 °C in the dark for 7 d using an Invisorb Spin Plant mini kit (Invitek, Berlin, Germany), following the manufacturer’s instructions. The internal transcribed spacer (ITS) regions and partial β–tubulin (tub2) genes were amplified using primer pairs ITS1/ITS4 (
PCR reactions were performed in 25 ml volumes (2.5 mM MgCl2, 1X PCR buffer, 0.2 mM dNTP, 0.2 mM of each primer and 2.5 U of Taq polymerase). The conditions for ITS and tub2 PCR amplifications were as described earlier (
Sequencing reactions were performed using CEQ DTCS Quick Start KitH (Beckman Coulter, American), following the manufacturer’s instructions, with the same PCR primers as above. Nucleotide sequences were determined using a CEQ 2000 XL capillary automated sequencer (Beckman Coulter).
Phylogenetic analyses
Contigs were subjected to BLAST searches of the NCBI GenBank database (https://www.ncbi.nlm.nih.gov/); published sequences of closely related species were retrieved. Alignments of the related genes (most up-to-date sequence regions deposited in the GenBank) were conducted online using MAFFT v 7.0 (https://mafft.cbrc.jp/alignment/server/index.html) (
The most parsimonious trees generated by MP analyses were identified by heuristic searches with a random addition sequence (1000); max trees were set to 200 and further evaluated by bootstrap analysis, retaining clades compatible with the 50% majority rule in the bootstrap consensus tree. The analysis was based on tree bisection reconnection branch swapping (TBR). The tree length (TL), consistency index (CI), retention index (RI), homoplasy index (HI) and rescaled consistency index (RC) were recorded for each dataset after tree generation.
The general-time-reversible (GTR) model for ML analyses was selected using the Akaike Information Criterion (AIC) in ModelTest v 3.7 (
For BI analyses, the most appropriate substitution models were also selected using the general-time-reversible model (GRT) with AIC in ModelTest v 3.7. BI was carried out with MrBayes using the Markov Chain Monte Carlo (MCMC) approach with 5,000,000 generations, to estimate posterior probabilities.
Results
Fungal isolation and sequence comparison
In total, 240 strains belonging to Ophiostomatales were obtained from PWN-infected galleries and pupal chambers of M. alternatus. The strains were sorted into six morphological groups (groups A–F in Table
Phylogenetic analyses
ITS and tub2 sequences were generated for 16 strains and deposited in GenBank (Table
The information of references sequences used for phylogenetic analyses in this study.
| Species | Strain No. | Host/insect | Country | Genbank No. | Reference | |
|---|---|---|---|---|---|---|
| ITS | β-tubulin | |||||
| Sporothrix abietina | CBS125.89 | Abies vejari | Mexico | AF484453 | KX590755 | de Beer et al. 2003 |
| S. aurorae | CMW19362 | Pinus eliottii | South Africa | DQ396796 | DQ396800 |
|
| S. bragantina | CBS 474.91 | Soil | Brazil | FN546965 | FN547387 |
|
| CBS 430.92 | Soil | Brazil | FN546964 | FN547386 |
|
|
| S. brasiliensis | Ss383 | Felis catus | Brazil | KP890194 | FN547387 |
|
| S. brunneoviolacea | CBS 124562 | Soil | Spain | FN546959 | FN547385 |
|
| CBS 124564 | Soil | Spain | FN546958 | FN547384 |
|
|
| S. dentifunda | CMW13016 | Quercus wood | Hungary | AY495434 | AY495445 |
|
| CMW13017 | Quercus wood | Poland | AY495435 | AY495446 |
|
|
| S. epigloea | CBS 573.63 | Tremella fusiformis | Argentina | KX590817 | KX590760 |
|
| S. eucalyptigena | CPC 24638 | Eucalyptus marginata | Western Australia | KR476721 | N/A |
|
| S. gemella | CMW23057 | Protea caffra | South Africa | DQ821560 | DQ821554 |
|
| S. inflata | CMW12529 | Soil | Canada | AY495428 | AY495438 |
|
| CMW12527 | wheat-field soil | Germany | AY495426 | AY495437 |
|
|
| S. nebularis | CMW27319 | Orthotomicus erosus | Spain | DQ674375 | N/A |
|
| CMW27900 | O. erosus | Spain | DQ674376 | N/A |
|
|
| S. pallida | CBS131.56 | Stemonitis fusca | Japan | EF127880 | EF139110 |
|
| CBS150.87 | S. fusca | Japan | EF127879 | EF139109 |
|
|
| S. palmiculminata | CMW23049 | Protea repens | South Africa | DQ316191 | DQ821543 |
|
| S. phasma | CMW20676 | P. laurifolia | South Africa | DQ316219 | DQ821541 |
|
| S. proteara | CMW1103 | P. caffra | South Africa | DQ316203 | DQ316165 |
|
| S. schenckii | MITS2474 | N/A | Mexico | KP132783 | N/A |
|
| CBS 938.72 | Human | Franch | KP017094 | N/A |
|
|
| S. fusiforis | CMW9968 | Populus nigra | Azerbaijan | AY280481 | AY280461 |
|
| S. lunata | CMW10563 | Carpinus betulus | Austria | AY280485 | AY280466 |
|
| S. narcissi | CBS138.50 | N/A | Canada | AY194510 | KX590765 |
|
| S. splendens | CMW872 | Protea repens | South Africa | DQ316215 | DQ316177 |
|
| S. stenoceras | CMW2524 | Acacia mearnsii | South Africa | AF484459 | AY280473 | de Beer et al. 2003 |
| CBS237.32 | pine pulp | Norway | AF484462 | N/A | de Beer et al. 2003 | |
| S. thermara | CMW38930 | Euphorbia ingens | South Africa | KR051115 | KR051103 |
|
| CMW38929 | E. ingens | South Africa | KR051114 | KR051102 |
|
|
| S. stylites | CMW14543 | Pine utility poles | Australia | EF127883 | EF139096 |
|
| Ophiostoma adjuncti | CMW135 | Pinus ponderosa | USA | AY546696 | N/A |
|
| O. allantosporum | CBS185.86 | P. pinaster | Europe | AY934506 | N/A |
|
| O. angusticollis | Zoq16 | N/A | N/A | EU109671 | N/A |
|
| CBS186.86 | Pinus banksiana | USA | AY924383 | KX590757 |
|
|
| O. bicolor | CBS492.77 | Picea glauca/Ips sp. | USA | DQ268604 | DQ268635 |
|
| O. candidum | CMW26484 | Eucalyptus cloeziana | South Africa | HM051409 | HM041874 |
|
| CMW26483 | E. cloeziana | South Africa | HM051408 | HM041873 |
|
|
| O. catonianum | C1084 | Pyrus | Italy | AF198243 | N/A |
|
| O. coronatum | CBS 497.77 | Pinus pinaster | Iberian Peninsula | AY924385 | KX590758 |
|
| O. cupulatum | C1194 | Pseudotsuga | USA | AF198230 | N/A |
|
| O. deltoideosporum | WIN(M)41 | N/A | N/A | EU879121 | N/A |
|
| O. fasciatum | UM56 | Pseudotsuga menziesii | CanadaCanada | EU913720 | EU913759 |
|
| O. floccosum | C01-021 | Girdled Picea rubens | Canada | AY194504 | N/A |
|
| C1086 | Soil | Sweden | AF198231 | N/A |
|
|
| O. fumeum | CMW26813 | Eucalyptus cloeziana | South Africa | HM051412 | HM041878 |
|
| CMW26818 | E. cloeziana | South Africa | HM051415 | HM041877 |
|
|
| O. fuscum | CMW23196 | Picea abies | Finland | HM031504 | HM031563 |
|
| O. himai ulmi | C1183 | Ulmus | India | AF198233 | N/A |
|
| C1306 | Ulmus | India | AF198234 | N/A |
|
|
| O. ips | CMW7075 | N/A | USA | AY546704 | N/A |
|
| CMW22843 | Orthotomicus erosus | N/A | DQ539549 | N/A |
|
|
| O. japonicum | YCC099 | N/A | N/A | GU134169 | N/A |
|
| O. kryptum | DAOM 229701 | Picea abies/Tetropium sp. | Austria | AY304436 | AY305685 | Jacobs and Kirisits 2013 |
| DAOM 229702 | Larix decidua/T. gabrieli | Austria | AY304434 | AY305686 | Jacobs and Kirisits 2013 | |
| K6/3/2 | Picea abies/Tetropium sp. | Austria | AY304428 | AY305687 | Jacobs and Kirisits 2013 | |
| O. minus | PIR 18S | N/A | N/A | AY934509 | N/A |
|
| CMW22802 | Dryocoetes autographus | N/A | DQ539507 | N/A | Romón et al. 2005 | |
| RJ-T144 | Tetropium sp. | Poland | AM943886 | N/A |
|
|
| CMW28117 | Picea abies/Tomicus minor | Russia | HM031497 | HM031535 |
|
|
| AU58.4 | Lodgepole pine | Canada | AF234834 | N/A |
|
|
| DAOM 212686 | N/A | Canada | AY304438 | AY305690 | Jacobs and Kirisits 2013 | |
| O. micans | CMW:38903 | Picea crassifolia | China | KU184432 | KU184303 |
|
| O. montium | CMW13221 | Pinus ponderosa/ Dendroctonus ponderosae | USA | AY546711 | N/A |
|
| CMW13222 | P. contorta/D. ponderosae | Canada | AY546712 | N/A |
|
|
| O. nigrocarpum | CMW 560 | Abies sp. | USA | AY280489 | AY280479 |
|
| CMW651 | Pseudotsuga menziesii | USA | AY280490 | AY280480 |
|
|
| O. nitidum | CMW:38907 | Picea crassifolia | China | KU184437 | KU184308 |
|
| O. novo ulmi | C1185 | Ulmus | Russia | AF198235 | N/A |
|
| C510 | Ulmus | USA | AF198236 | N/A |
|
|
| O. olgensis | CXY1404 | Larix gmelini/Ips subelongatus | China | KU551299 | KU882938 |
|
| CXY1405 | L. gmelini/I. subelongatus | China | KU551300 | KU882939 |
|
|
| CXY1410 | L. gmelini/I. subelongatus | China | KU551303 | KU882942 |
|
|
| O. pallidulum | CMW23279 | Pinus sylvestris/Hylastes brunneus | Finland | HM031509 | N/A |
|
| CMW23278 | P. sylvestris/ H. brunneus | Finland | HM031510 | HM031566 |
|
|
| O. piceae | C1087 | N/A | Germany | AF198226 | N/A |
|
| C1246 | Pseudotsuga | USA | AF198227 | N/A |
|
|
| O. pseudotsugae | 92-634/302/6 | Pinus menziesii/Dendroctonus frontalis | Canada | AY542502 | AY548744 |
|
| D48/3 | N/A | Canada | AY542501 | AY542511 |
|
|
| O. proteasedis | CMW 28601 | Protea caffra | Zambia | EU660449 | EU660464 |
|
| O. pulvinisporum | CMW9022 | Pinus pseudostrobus/Dendroctonus mexicanus | Mexico | AY546714 | DQ296100 |
|
| O. qinghaiense | CMW:38902 | Picea crassifolia | China | KU184445 | KU184316 |
|
| O. querci | C970 | Quercus | United Kingdom | AF198239 | N/A |
|
| C969 | Quercus | United Kingdom | AF198238 | N/A |
|
|
| C1085 | Fagus | Germany | AF198237 | N/A |
|
|
| O. rostrocoronatum | CBS434.77 | Woodpulp | USA | AY194509 | KX590771 |
|
| O. saponiodorum | CMW29497 | Picea abies/Ips typographus | Finland | HM031507 | HM031571 |
|
| CMW28135 | P. abies | Russia | HM031508 | N/A |
|
|
| O. sejunctum | Ophi 1B | N/A | N/A | AY934520 | N/A |
|
| Ophi 1A | N/A | N/A | AY934519 | N/A |
|
|
| O. setosum | AU160-38 | Pseutotsugae menziesii | North America | AF128929 | N/A |
|
| CMW12378 | Tsuga sp. | China | FJ430485 | FJ430515 |
|
|
| O. tenellum | CBS189.86 | Pinus banksiana | USA | AY934523 | KX590772 |
|
| O. tetropii | C00-027a | Tetropium fuscum | Canada | AY194482 | NA |
|
| C00-003 | T. fuscum | Canada | AY194485 | AY305701 |
|
|
| O. ulmi | C1182 | Ulmus | Netherlands | AF198232 | N/A |
|
| Graphilbum crescericum | CMW 22829 | Hylastes ater | Spain | DQ539535 | N/A |
|
| Gra. fragrans | C1224 | Pinus sylvestris | Sweden | AF198248 | N/A |
|
| Gra. microcarpum | YCC612 | Japanese larch logs | Japan | GU134170 | N/A |
|
| Gra. rectangulosporium | MAFF 238951 | N/A | Japan | AB242825 | N/A |
|
| Raffaelea canadensis | CBS 168.66 | N/A | N/A | GQ225699 | N/A |
|
| Leptographium lundbergii | DAOM 64746 | N/A | N/A | EU879151 | AY534943 |
|
| L. truncatum | WIN(M)1435 | Pinus taeda | South Africa | AY935626 | N/A |
|
For each phylogenetic tree, MP, ML and BI analyses yielded trees with very similar topologies. Phylograms, generated by the MP analysis, are presented for all the datasets, with nodal support obtained from ML indicated at the nodes (Figure
Phylograms of fungal associates of pine infected by PWN and Monochamus alternatus in China. The phylograms were generated after MP analysis of the ITS1–5.8S–ITS2 rDNA and partial tub2 sequences. Novel sequences obtained in the current study are indicated in bold type. MP bootstrap values (10,000 replicates) and ML bootstrap support values (1000 replicates) (normal type) above 70% are indicated at the nodes. Values below 70% are indicated by asterisk (*). Posterior probabilities (above 90%) obtained from BI are indicated by bold lines at the relevant branching points. Scale bar, total nucleotide differences between taxa; ML, maximum likelihood; MP, maximum parsimony; BI, Bayesian inference.
According to the ITS sequence analysis, strains of the morphological group A nested in the Sporothrix lineage, as defined by
Phylogenetic inferences based on tub2 sequences revealed that clade A, B and D strains formed three well-supported independent clades within the Sporothrix and Ophiostoma lineages, respectively. Clade C strains nested within the well-supported O. ips clade (Suppl. material
Considering morphological differences, strains in groups A, B and D represent three undescribed species of Sporothrix or Ophiostoma. We concluded that group C strains belong to O. ips; group E and F strains clustered together with the well-supported Graphilbum rectangulosporium and O. deltoideosporum clades, respectively. However, because of a limited number of strains, further analysis of this potential species will need to be postponed until a sufficient amount of material obtained.
Taxonomy
Based on the phylogenetic signals of the ITS and tub2 and morphological characteristics, all strains analysed in the current study were assigned to six different groups (A–F). They represent one known species, O. ips (
Sporothrix zhejiangensis , sp. nov.
Etymology
The epithet reflects Zhejiang Province in China where the species was first collected.
Type
CHINA, Zhejiang, Yuyao City, from Monochamus alternatus gallery in Pinus massoniana infested by numerous PWN, November 2012, collected by Q Lu and YY Lun, culture ex-holotype MUCL 55183 = CFCC52165 = CXY1614.
Description
Sexual morph perithecial: Perithecia occasional on 2% MEA, emerging from the superficial mycelium or partly iμmersed, with a globose base, (75–)80–108(–120) μm in diameter, with some basal hyphal ornamentation, black; extending progressively into a straight, brown to black neck, (127–)156–550(–631) μm long, (26–)32–58.5(–65) μm wide at the base, (7–)7.5–10.7(–12) μm wide at the apex; ending in a crown of hyaline, (6–)9–19.5(–24) μm long ostiolar hyphae; ascospores reniform in side view, without sheath, aseptate, hyaline, (2–)2.2–3.4(–4) × (0.6–)0.74–2(–2.5) μm.
Asexual morph: pesotum-like and sporothrix-like.
Pesotum-like: Conidiophores macronematous, synnematous, abundant in 2% MEA. Synnemata occurring singly, enlarging towards both the apex and the base, dark brown at base, becoming paler toward the apex, (100–)120–260(–290) μm long including the conidiogenous apparatus, (56–)63–145(–158) μm wide at base, rhizoids present; conidiogenous cells (7–)9.5–29(–45.5) × 1–2(–1.7) μm; conidia hyaline, aseptate, single-celled, smooth, cylindrical or obovoid, (2–)2.5–4.8(–6) × (0.5–)0.8–2.1(–2.6) μm.
Sporothrix-like: Conidiophores micronematous, single on aerial mycelia, unbranched, (4.5–)9.6–31.5(–51.5) × (1.0–)1.5–2(–2.4) μm; conidia hyaline, smooth, aseptate, ellipsoid to ovoid, (2.5–)3–4.8(–5) × (0.7–)1–2.1(–2.5) μm.
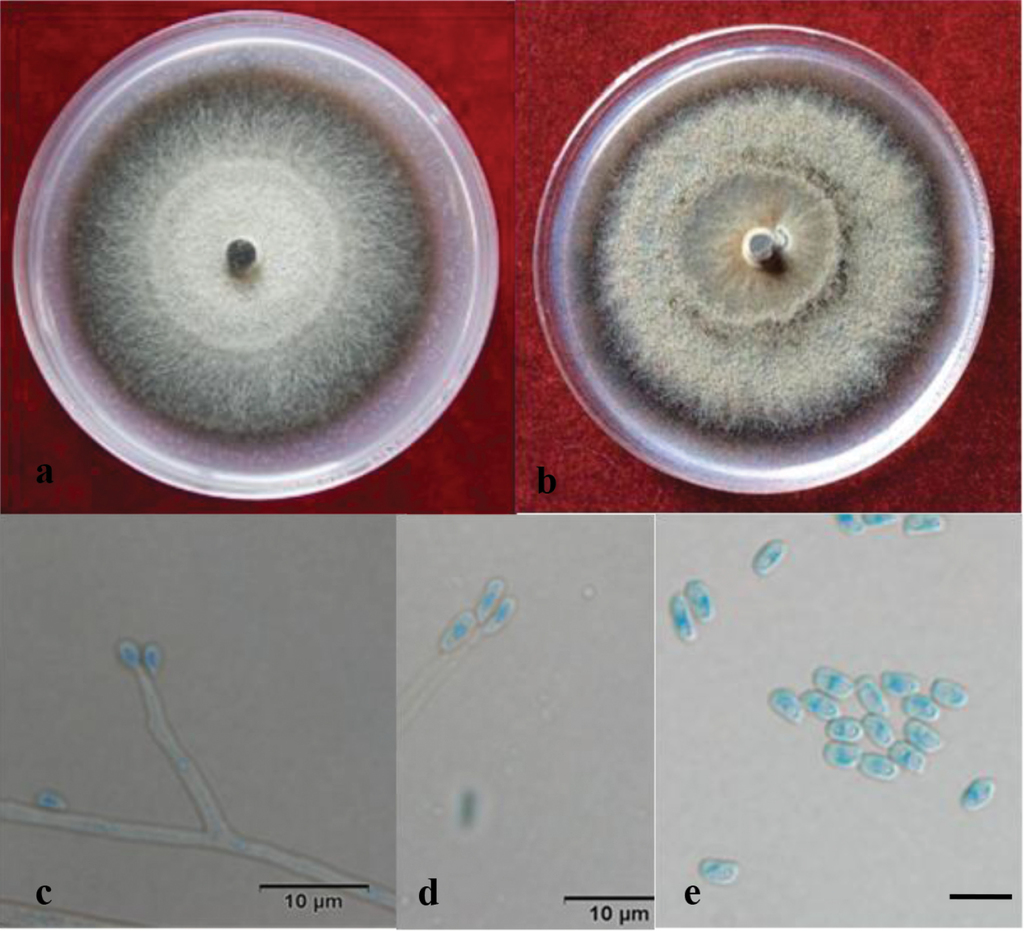
Light micrographs of Sporothrix zhejiangensis. a–c Growth on 2% MEA and 2% PDA, 2 weeks after inoculation d Occasionally observed ostiolar hyphae (scale bar, 20 μm) e–f Perithecium (scale bar, 20 μm) g Pesotum-like anamorph, rhizoid, conidiophores, conidiogenous apparatus (scale bar, 20 μm), and conidia (bottom right corner) (scale bar, 10 μm) h, i Reniform ascospores without sheaths (scale bar, 10 μm) j–lSporothrix-like anamorph, conidiophores, and conidia (scale bar, 10 μm).
Culture characteristics
Colonies on 2% MEA medium are white, with colony edge thinning radially. Hyphae are superficial on agar. Diameter reaches 50 μm in the dark after 8 d at 25 °C, able to grow at 5 °C and 40 °C, with the optimal growth temperature of 30 °C. Growth characteristics on PDA medium are similar.
Habitat and distribution
Galleries of Monochamus alternatus in Pinus massoniana infested by PWN; known hitherto from Zhejiang Province, China.
Additional specimens examined
CHINA, Zhejiang, Yuyao City, from Monochamus alternatus galleries in Pinus massoniana infested by PWN, November 2012, collected by Q Lu and YY Lun, MUCL 55181 = CFCC 52167 = CXY1612, MUCL 55182 = CFCC 52164 = CXY1613, MUCL 55184 = CFCC 52166 = CXY1615.
Note
Sporothrix zhejiangensis is characterised by a sexual and two asexual forms (pesotum-like and sporothrix-like). It is phylogenetically related to S. nebulare, S. eucalyptigena and S. epigloea (Figure
Sporothrix eucalyptigena and S. epigloea produce perithecia and ascospores similar to those of S. zhejiangensis (
Sporothrix zhejiangensis is also closely related to S. bragantina and S. thermara (Figure
Ophiostoma album , sp. nov.
Etymology
The epithet reflects the white colour of the colonies.
Type
CHINA, Zhejiang, Yuyao City, from Monochamus alternatus gallery of Pinus massoniana infested by numerous PWN, November 2012, collected by Q Lu and YY Lun, culture ex-holotype MUCL 55189 = CFCC 52168 = CXY1622.
Description
Sexual form: Unknown. Asexual form: Hyalorhinocladiella-like. Conidiogenous cells micronematous, (4.2–)9.5–16.5(–20.5) × (0.5–)1–2(–2.5) μm; conidia hyaline, single-celled, aseptate, clavate or fusiform obovoid with pointed bases and (occasionally) rounded apices, slightly curved at the base (4–)4.2–14.5(–18) × (0.5–)1–2(–2.3) μm.
Culture characteristics
Colonies on 2% MEA white, with the mycelium edge thinning radially; Hyphae are superficial on agar, sporulation weak. Colonies slowly growing, reaching 18.5 μm in diameter at 8 d at 25 °C, able to grow at 40 °C but not at 5 °C, with the optimal growth temperature of 35 °C. Growth characteristics on PDA culture medium are similar but the growth rate is slower than on MEA.
Habitat and distribution
Galleries of Monochamus alternatus in Pinus massoniana, infested by PWN, in Zhejiang Province, China.
Additional specimens examined
CHINA, Zhejiang, Yuyao City, from Monochamus alternatus galleries of Pinus massoniana infested by numerous PWN, November 2012, collected by Q Lu and YY Lun, MUCL 55190 = CFCC 52169 = CXY1642, CXY1643 = CFCC 52170.
Note
Ophiostoma album only known in its asexual hyalorhinocladiella-like form. According to both ITS and tub2 based phylogenetic analysis, it is closely related to O. kryptum and O. olgensis in the O. minus complex (Figure
Ophiostoma massoniana , sp. nov.
Etymology
The epithet reflects the host tree, Pinus massoniana.
Type
CHINA, Zhejiang Province, Fuyang City, from Monochamus alternatus gallery in Pinus massoniana infested by numerous PWN, November 2012, collected by Q Lu and YY Lun, culture ex-holotype, MUCL 55179 = CFCC 51648 = CXY1610.
Description
Sexual form: Unknown. Asexual form: Hyalorhinocladiella-like. Conidiophores abundant, single, borne on aerial hyphae, (3.3–)10.5–27.5(–42.5) × (0.7–)1.3–2.0(–2.7) μm; conidia hyaline, single-celled, aseptate, obovoid or globose with pointed bases and rounded apices, (2–)2.2–3.9(–5) × (0.5–)0.7–1.7(–2) μm.
Culture characteristics
Colonies on 2% MEA brown, the marginal hyphae sparse and radiating; some white mycelium produced early during growth that becomes black after 3–5 d. Colonies slowly growing, reaching 37.5 μm in diameter over 8 d at 25 °C, able to grow at 5 °C and 40 °C, with an optimal growth temperature of 30 °C; sporulation weak. On PDA culture medium, the colonies are dark brown; the mycelium is white, long and dense, with a daily growth of 4 μm at 25 °C.
Habitat and distribution
Galleries of Monochamus alternatus in Pinus massoniana infested by PWN, in Zhejiang Province, China.
Additional specimens examined
CHINA, Zhejiang Province, Yuyao City, from Monochamus alternatus galleries in Pinus massoniana infested by numerous PWN, November 2012, collected by Q Lu and YY Lun, MUCL 55180 = CFCC 51649 = CXY1611.
Note
Ophiostoma massoniana, only known by its asexual, hyalorhinocladiella-like state, does not cluster in any of the 10 species complexes defined by
Discussion
In the current study, six ophiostomatoid species were found associated with pines infected by M. alternatus and PWN in the eastern provinces of Shandong and Zhejiang in China: O. ips, the newly described S. zhejiangensis, O. album, O. massoniana and two species whose identities are uncertain; O. cf. deltoideosporum and Gra. cf. rectangulosporium. Ophiostoma ips was the most frequently isolated species, accounting for over 90% of all Ophiostomatales strains.
Ophiostoma ips was originally reported in association with bark beetles infecting pines in south-eastern North America (
In China, O. ips was reportedly associated with P. massoniana infected by PWN (
Ophiostoma ips appears to have travelled long-distances in wood materials presumably originating from North America (
Members of Sporothrix are reportedly associated with a wide range of habitats (
Sporothrix zhejiangensis forms an independent lineage according to both ITS and tub2 based on phylogenetic inferences. It is closely related to S. nebulare, S. eucalyptigena, S. epigloea, S. bragantina and S. thermara (
Although S. zhejiangensis is unrelated to S. fusiforis, S. lunata and S. stenoceras (Figure
In the current study, S. zhejiangensis was notably different from Sporothrix sp. 1 and Sporothrix sp. 2 (
According to ITS phylogeny analysis, Ophiostoma album is related to O. olgensis (
The O. minus complex currently includes O. minus, O. pseudotsugae, O. allantosporum, O. kryptum and O. olgensis (
According to both ITS and tub2 phylogenetic trees, O. massoniana forms a separated well-supported clade (Figure
Conclusions
In the current study, a relatively large number of ophiostomatoid fungal species associated with B. xylophilus and M. alternatus in Shandong and Zhejiang Provinces in China was identified. Three novel species, O. album, O. massoniana and S. zhejiangensis were discovered and described. Fourteen additional provinces in China are currently also listed as PWN epidemic areas (
Acknowledgments
This work was supported by the National Key R&D Programme of China (2017YFD0600103) and the National Natural Science Foundation of China (Project No.: 31770682). Cony Decock gratefully acknowledges the financial support received from the Belgian State (Belgian Federal Science Policy through the BCCMTM research programme). We are grateful to Professor Yuichi Yamaoka and Hugo Madrid for their invaluable suggestions to improve the manuscript.
References
- Aghayeva DN, Wingfield MJ, Kirisits T, Wingfield BD (2005) Ophiostoma dentifundum sp. nov. from oak in europe, characterized using molecular phylogenetic data and morphology. Mycological Research 109(Pt 10): 1127. https://doi.org/10.3767/003158509X468038
- Aghayeva DN, Wingfield MJ, de Beer ZW, Kirisits T (2004) Two new Ophiostoma species with Sporothrix anamorphs from Austria and Azerbaijan. Mycologia 96(4): 866–878. https://doi.org/10.2307/3762119
- Araujo ML, Rodrigues AM, Fernandes GF, Camargo ZP, Hoog GS (2015) Human Sporotrichosis beyond the epidemic front reveals classical transmission types in espírito santo, brazil. Mycoses 58(8): 485. https://doi.org/10.1111/myc.12346
- Benade E, Wingfield MJ, Van Wyk PS (1995) Conidium development in the Hyalorhinocladiella anamorph of Ophiostoma ips. Mycologia 87(3): 298–303. https://doi.org/10.2307/3760826
- Chang R, Duong TA, Taerum SJ, Wingfield MJ, Zhou X, De Z B (2017) Ophiostomatoid fungi associated with conifer-infesting beetles and their phoretic mites in yunnan, China. Mycokeys 28(28): 19–64. https://doi.org/10.3897/mycokeys.28.21758
- Crous PW, Wingfield MJ, Guarro J, Hernández-Restrepo M, Sutton DA, Acharya K, Barber PA, Boekhout T, Dimitrov RA, Dueñas M, Dutta AK, Gené J, Gouliamova DE, Groenewald M, Lombard L, Morozova OV, Sarkar J, Smith MT, Stchigel AM, Wiederhold NP, Alexandrova AV, Antelmi I, Armengol J, Barnes I, Cano-Lira JF, Castañeda Ruiz RF, Contu M, Courtecuisse PR, da Silveira AL, Decock CA, de Goes A, Edathodu J, Ercole E, Firmino AC, Fourie A, Fournier J, Furtado EL, Geering AD, Gershenzon J, Giraldo A, Gramaje D, Hammerbacher A, He XL, Haryadi D, Khemmuk W, Kovalenko AE, Krawczynski R, Laich F, Lechat C, Lopes UP, Madrid H, Malysheva EF, Marín-Felix Y, Martín MP, Mostert L, Nigro F, Pereira OL, Picillo B, Pinho DB, Popov ES, Rodas Peláez CA, Rooney-Latham S, Sandoval-Denis M, Shivas RG, Silva V, Stoilova-Disheva MM, Telleria MT, Ullah C, Unsicker SB, van der Merwe NA, Vizzini A, Wagner HG, Wong PT, Wood AR, Groenewald JZ (2015) Fungal Planet description sheets: 320–370. Persoonia 34: 167–266. https://doi.org/10.3767/003158515X688433
- de Beer ZW, Wingfield MJ (2013) Emerging lineages in the Ophiostomatales. In: Seifert KA, de Beer ZW, Wingfield MJ (Eds) The Ophiostomatoid Fungi: Expanding Frontiers.CBS-KNAW Fungal Biodiversity Centre, Utrecht, The Netherlands, 21–46.
- de Beer ZW, Duong TA, Wingfield MJ (2016) The divorce of Sporothrix and Ophiostoma: solution to a problematic relationship. Studies in Mycology 83: 165–191. https://doi.org/10.1016/j.simyco.2016.07.001
- de Beer ZW, Seifert KA, Wingfield MJ (2013) The ophiostomatoid fungi: Their dual position in the sordariomycetes. In: Seifert KA, de Beer ZW, Wingfield MJ (Eds) The Ophiostomatoid Fungi: Expanding Frontiers.CBS-KNAW Fungal Biodiversity Centre, Utrecht, The Netherlands, 1–19.
- de Hoog GS (1974) The genera Blastobotrys, Sporothrix, Calcarisporium and Calcarisporiella gen. nov. Studies in Mycology 7: 1–84.
- de Meyer EM, de Beer ZW, Summerbell RC, Moharram AM, de Hoog GS, Vismer HF, Wingfield MJ (2008) Taxonomy and phylogeny of new wood- and soil-inhabiting species in the Ophiostoma stenoceras-Sporothrix schenckii complex. Mycologia 100(4): 647–661. https://doi.org/10.3852/07-157R
- Evans H, McNamara DG, Braasch H, Chadoeuf J, Magnusson C (1996) Pest Risk Analysis (PRA) for the territories of the European Union (as PRA Area) on Bursaphelenchus xylophilus and its vectors in the genus Monochamus. European and Mediterranean Plant Protection Organization Bulletin 26(2): 199–249. https://doi.org/10.1111/j.1365-2338.1996.tb00594.x
- Francois R, Wilhelm DBZ, Dreyer LL, Renate Z, Crous P, Wingfield MJ (2006) Multi-gene phylogeny for Ophiostoma spp. reveals two new species from Protea infructescences. Studies in Mycology 55(1): 199–212.
- Futai K (2013) Pine wood nematode, Bursaphelenchus xylophilus. Annual Review of Phytopathology 51: 61–83. https://doi.org/10.1146/annurev-phyto-081211-172910
- Glass NL, Donaldson GC (1995) Development of primer sets designed for use with the PCR to amplify conserved genes from filamentous ascomycetes. Applied and Environmental Microbiology 61(4): 1323–1330.
- Gorton C, Kim SH, Henricot B, Webber J, Breuil C (2004) Phylogenetic analysis of the bluestain fungus Ophiostoma minus based on partial ITS rDNA and β-tubulin gene sequences. Mycological Research 108: 759–765. https://doi.org/10.1017/S0953756204000012
- Grobbelaar JW, Aghayeva DN, Beer ZWD, Bloomer P, Wingfield MJ, Wingfield BD (2009) Delimitation of Ophiostoma quercus, and its synonyms using multiple gene phylogenies. Mycological Progress 8(3): 221–236. https://doi.org/10.1007/s11557-009-0594-4
- Guidon S, Gascuel O (2003) A simple, fast, and accurate algorithm to estimate large phylogenies by maximum likelihood. Syst Biol 52: 696–704. https://doi.org/10.1080/10635150390235520
- Harrington TC, Mcnew D, Steimel J, Hofstra D, Farrell R (2001) Phylogeny and taxonomy of the Ophiostoma piceae complex and the dutch elm disease fungi. Mycologia 93(1): 111–136. https://doi.org/10.2307/3761610
- Hsiau PTW (1996) The taxonomy and phylogeny of the mycangial fungi from Dendroctonus brevicomis and D. frontalis (Coleoptera: Scolytidae). PhD Thesis, Iowa State University, Ames, Iowa.
- Hausner G, Iranpour M, Kim JJ, Breuil C, Davis CN, Gibb EA, Loewen PC, Hopkin AA (2005) Fungi vectored by the introduced bark beetle Tomicus piniperda in Ontario, Canada, and comments on the taxonomy of Leptographium lundbergii, Leptographium terebrantis, Leptographium truncatum, and Leptographium wingfieldii. Canadian Journal of Botany 83(10): 1222–1237. https://doi.org/10.1139/b05-095
- Hyun MW, Kim JH, Suh DY, Lee SK, Kim SH (2007) Fungi isolated from pine wood nematode, its vector Japanese pine sawyer, and the nematode-infected Japanese black pine wood in Korea. Mycobiology 35(3): 159–161. https://doi.org/10.4489/MYCO.2007.35.3.159
- Irinyi L, Serena C, Garcia-Hermoso D, Arabatzis M, Desnos-Ollivier M, Vu D, Cardinal G, Arthur I, Normand AC, Giraldo A, da Cunha KC, Sandoval-Denis M, Hendrickx M, Nishikaku AS, de Azevedo Melo AS, Merseguel KB, Khan A, Parente Rocha JA, Sampaio P, da Silva Briones MR, Carmona e Ferreira R, de Medeiros Muniz M, Castanon-Olivares LR, Estrada-Barcenas D, Cassagne C, Mary C, Duan SY, Kong F, Sun AY, Zeng X, Zhao Z, Gantois N, Botterel F, Robbertse B, Schoch C, Gams W, Ellis D, Halliday C, Chen S, Sorrell TC, Piarroux R, Colombo AL, Pais C, de Hoog S, Zancope-Oliveira RM, Taylor ML, Toriello C, de Almeida Soares CM, Delhaes L, Stubbe D, Dromer F, Ranque S, Guarro J, Cano-Lira JF, Robert V, Velegraki A, Meyer W (2015) International society of human and animal mycology (isham)-its reference DNA barcoding database-the quality controlled standard tool for routine identification of human and animal pathogenic fungi. Medical Mycology 53(4): 313. https://doi.org/10.1093/mmy/myv008
- Ja VDL, Six DL, De Beer WZ, Wingfield MJ, Roux J (2016) Novel ophiostomatalean fungi from galleries of Cyrtogenius Africus (Scolytinae) infesting dying Euphorbia Ingens. Antonie Van Leeuwenhoek 109(4): 589–601. https://doi.org/10.1007/s10482-016-0661-1
- Jacobs K, Kirisits T (2003) Ophiostoma kryptum sp. nov. from Larix decidua and Picea abies in Europe, similar to O. minus. Mycological Research 107(10): 1231–1242. https://doi.org/10.1017/S0953756203008402
- Jacobs K, Seifert KA, Harrison KJ, Kirisits T (2003) Identity and phylogenetic relationships of ophiostomatoid fungi associated with invasive and native Tetropium species (coleoptera: cerambycidae) in Atlantic Canada. Canadian Journal of Botany 81(4): 316–329. https://doi.org/10.1139/b03-025
- Jacobs K, Wingfield MJ (2001) Leptographium Species: tree pathogens, insect associates and agents of blue-stain. American Phytopathological Society Press, St Paul, MN.
- Jankowiak R, KolařÍk M (2010) Diversity and pathogenicity of ophiostomatoid fungi associated with Tetropium species (coleoptera: cerambycidae) colonizing Picea abies in Poland. Folia Microbiologica 55(2): 145–154. Https://doi.org/10.1007/s12223-010-0022-9
- Jung J, Han H, Ryu SH, Kim W (2010) Microsatellite variation in the pinewood nematode, Bursaphelenchus xylophilus (Steiner and Buhrer) Nickle in South Korea. Genes & Genomics 32(2): 151–158. https://doi.org/10.1007/s13258-009-0842-7
- Katoh K, Standley DM (2013) MAFFT multiple sequence alignment software version 7: improvements in performance and usability. Molecular biology and evolution 30(4): 772–780. https://doi.org/10.1093/molbev/mst010t
- Kwon-Chung KJ, Bennett JE (1992) Medical mycology. Lea & Febiger, Philadelphia, 866 pp. https://doi.org/10.1590/S0036-46651992000600018
- Kyunghee K, Youngjoon C, Sangtae S, Hyeondong S (2009) Raffaelea quercus-mongolicae sp. nov. associated with Platypus koryoensis on oak in Korea. Mycotaxon 110(8): 189–197.
- Linnakoski R, de Beer ZW, Ahtiainen J, Sidorov E, Niemelä P, Pappinen A, Wingfield MJ (2010) Ophiostoma spp. associated with pine-and spruce-infesting bark beetles in Finland and Russia. Persoonia: Molecular Phylogeny and Evolution of Fungi 25: 72. https://doi.org/10.3767/003158510X550845
- Lu M, Zhou XD, de Beer ZW, Wingfield MJ, Sun JH (2009) Ophiostomatoid fungi associated with the invasive pine-infesting bark beetle, Dendroctonus valens, in China. Fungal Divers 38: 133–45.
- Madrid H, Cano J, Gene J, Bonifaz A, Toriello C (2009) Sporothrix globosa, a pathogenic fungus with widespread geographical distribution. Rev Iberoam Micol 26: 218–222. https://doi.org/10.1016/j.riam.2009.02.005
- Madrid H, Gene J, Cano J, Silvera C, Guarro J (2010) Sporothrix brunneoviolacea and Sporothrix dimorphospora, two new members of the Ophiostoma stenoceras-Sporothrix schenckii complex. Mycologia 102(5): 1193–1203. https://doi.org/10.3852/09-320
- Maehara N, Futai K (1997) Effect of fungal interactions on the numbers of the pinewood nematode, Bursaphelenchus xylophilus (Nematoda: Aphelenchoididae), carried by the Japanese pine sawyer, Monochamus alternatus (Coleoptera: Cerambycidae). Fundamental and applied Nematology 20(6): 611–618.
- Mamiya Y, Shoji T (2009) Pathogenicity of the pinewood nematode, Bursaphelenchus xylophilus, to Japanese larch, Larix kaempferi, seedlings. J. Nematol 41: 157–162.
- Mota M, Vieira P (2008) Pine wilt disease: a worldwide threat to forest ecosystems. M M. Mota. Springer, Dordrecht, 428 pp.
- Massoumi AS, Kim JJ, Humble LM, Uzunovic A, Breuil C (2007) Ophiostomatoid fungi associated with the Northern Spruce engraver, Ips perturbatus, in Western Canada. Antonie Van Leeuwenhoek 91(1): 19–34. https://doi.org/10.1007/s10482-006-9092-8
- Mota MM, Vieira P (2008) Pine Wilt Disease: A Worldwide Threat to Forest Ecosystems. Springer, Netherlands. https://doi.org/10.1007/978-1-4020-8455-3
- Mullineux T, Hausner G (2009) Evolution of rdna its1 and ITS2 sequences and RNA secondary structures within members of the fungal genera Grosmannia and Leptographium. Fungal Genetics & Biology Fg & B, 46(11): 855. https://doi.org/10.1016/j.fgb.2009.08.001
- Nkuekam GK, Beer ZWD, Wingfield MJ, Roux J (2012) A diverse assemblage of Ophiostoma species, including two new taxa on Eucalypt trees in South Africa. Mycological Progress, 11(2): 515–533. https://doi.org/10.1007/s11557-011-0767-9
- Ohtaka N, Masuya H, Yamaoka Y, Kaneko S (2006) Two new Ophiostoma species lacking conidial states isolated from bark beetles and bark beetle-infested Abies species in Japan. Revue Canadienne De Botanique, 84(84): 282–293. https://doi.org/10.1139/B05-164
- Pérez-Vera OA, Cárdenas-Soriano E, Alvarado-Rosales D, Cibrián-Tovar D, Equihua-Martínez A (2011) Histopathology of Hartweg pine (Pinus hartwegii lindl.) innoculated with three ophiostomatoid fungi. Revista Chapingo Serie Ciencias Forestales Y Del Ambiente 17(1): 91–102. https://doi.org/10.5154/r.rchscfa.2010.03.006
- Pfenning L, Oberwinkler F (1993) Ophiostoma bragantinum n. sp., a possible teleomorph of Sporothrix inflata, found in Brazil. Mycotaxon 46: 381–385.
- Plattner A, Jaejin K, Reid J, Hausner G, Youngwoon L, Yamaoka Y, Breuil C (2009) Resolving taxonomic and phylogenetic incongruence within species Ceratocystiopsis minuta. Mycologia, 101(6): 878. https://doi.org/10.3852/08-132
- Posada D, Crandall KA (1998) MODELTEST: testing the model of DNA substitution. Bioinformatics 14: 817–818. https://doi.org/10.1093/bioinformatics/14.9.817
- Rane KK, Tattar TA (1987) Pathogenicity of blue-stain fungi associated with Dendroctonus terebrans. Plant Dis 71: 8798–8883. https://doi.org/10.1094/PD-71-0879
- Rayner RW (1970) A mycological colour chart. Commonwealth Mycological Institute and British Mycological Society.
- Robbins K (1982) Distribution of the pinewood nematode in the United States. In: Appleby JE, Malek RB (Eds) Proceedings of the national pine wilt disease workshop.III. Natural History Survey, Champaign, IL, USA, 3–6.
- Roets F, Wingfield MJ, Crous PW, Dreyer LL, Savolainen V, Dreyer LL (2009) Fungal radiation in the cape floristic region: an analysis based on Gondwanamyces and Ophiostoma. Molecular Phylogenetics & Evolution 51(1): 111–119. https://doi.org/10.1016/j.ympev.2008.05.041
- Roets F, de Beer ZW, Wingfield MJ, Crous PW, Dreyer LL (2008) Ophiostoma gemellus and Sporothrix variecibatus from mites infesting Protea infructescences in South Africa. Mycologia 100: 496–510. https://doi.org/10.3852/07-181R
- Roets F, de Beer ZW, Dreyer LL, Zipfel R, Crous PW, Wingfield MJ (2006) Multi-gene phylogeny for Ophiostoma spp. reveals two new species from Protea infructescences. Stud Mycol 55: 199–212. https://doi.org/10.3114/sim.55.1.199
- Romón P, Zhou X, Iturrondobeitia JC, Wingfield MJ, Goldarazena A (2007) Ophiostoma species (ascomycetes: ophiostomatales) associated with bark beetles (coleoptera: Scolytinae) colonizing Pinus radiata in Northern Spain. Canadian Journal of Microbiology 53(6): 756–767. https://doi.org/10.1139/W07-001
- Romón P, de Beer ZW, Zhou XD, Duong TA, Wingfield BD, Wingfield MJ (1900) Multigene phylogenies of Ophiostomataceae associated with Monterey pine bark beetles in Spain reveal three new fungal species. Mycologia 106(1): 119–32. https://doi.org/10.3852/13-073
- Rumbold CT (1931) Two blue-staining fungi associated with bark beetle infestation of pines. Journal of Agricultural Research 43(10): 847–873.
- Ryss A, Vieira P, Mota M, Kulinich O (2005) A synopsis of the genus Bursaphelenchus Fuchs, 1937 (Aphelenchida: Parasitaphelenchidae) with keys to species. Nematology 7(3): 393–458. https://doi.org/10.1163/156854105774355581
- Seifert KA, Webber JF, Wingfield MJ (1993) Methods for studying species of Ophiostoma and Ceratocystis. In: Wingfield MJ, Seifert KA, Webber JF, eds. Ceratocystis and Ophiostoma: taxonomy, ecology and pathogenicity. Minnesota, U.S.A.: The American Phytopathological Society Press, 255–259.
- State Forestry Administration of the People’s Republic of China (2018) Announcement of State Forestry Administration of the People’s Republic of China No. 1, 2018.
- Steiner G, Buhrer EM (1934) Aphelenchoides Xylophilus N. Sp., a Nematode Associated with Blue-Stain and Other Fungi in Timber. Journal of Agricultural Research 48(10): 949–951.
- Suh DY, Hyun MW, Kim JJ, Son SY, Kim SH (2013) http://dx.doi.org/10.5941/MYCO.2013. 41.1.59
- Swofford DL (2003) PAUP*: phylogenetic analysis using parsimony, version 4.0 b10.
- Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S (2011) MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol 28: 2731–2739. https://doi.org/10.1093/molbev/msr121
- Upadhyay HP (1981) A monograph of Ceratocystis and Ceratocystiopsis. The University of Georgia Press, Athens, GA, USA, 95 pp.
- Uzunovic A, Seifert KA, Kim SH, Breuil C (2000) Ophiostoma setosum, a common sapwood staining fungus from western North America, a new species of the Ophiostoma piceae, complex. Mycological Research 104(4): 486–494. https://doi.org/10.1017/S0953756299001446
- Van der Linde JA, Six DL, de Beer WZ, Wingfield MJ, Roux J (2016) Novel ophiostomatalean fungi from galleries of Cyrtogenius africus (Scolytinae) infesting dying Euphorbia ingens. Antonie van Leeuwenhoek 109(4): 589–601. https://doi.org/10.1007/s10482-016-0661-1
- Villarreal M, Rubio V, Mtde T, Arenal F (2005) A new Ophiostoma species isolated from Pinus pinaster in the Iberian peninsula. Mycotaxon 92(3): 259–268.
- Wang HM, Lu Q, Meng XJ, Liu XW, Decock C, Zhang XY (2016) Ophiostoma olgensis, a new species associated with Larix spp. and Ips subelongatus in northern China. Phytotaxa 282(4): 282–290. https://doi.org/10.11646/phytotaxa.282.4.5
- White TJ, Bruns T, Lee SJWT, Taylor JL (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. PCR protocols: a guide to methods and applications 18(1): 315–322. https://doi.org/10.1016/B978-0-12-372180-8.50042-1
- Wingfield MJ (1987) Fungi associated with the pinewood nematode, Bursaphelenchus xylophilus, and cerambycid beetles in Wisconsin. Mycologia 79: 325–328. https://doi.org/10.2307/3807667
- Yamaoka Y, Chung WH, Masuya H, Hizai M (2009) Constant association of ophiostomatoid fungi with the bark beetle Ips subelongatus, invading Japanese larch logs. Mycoscience, 50(3): 165–172. https://doi.org/10.1007/s10267-008-0468-7
- Yamaoka Y, Takahashi I, Iguchi K (2000) Virulence of ophiostomatoid fungi associated with the spruce bark beetle Ips typographus f. japonicus in Yezo spruce. Journal of Forest Research 5(2): 87–94. https://doi.org/10.1007/BF02762525
- Yan DH, Lu Q, Zhang XY (2003) Pine Wilt Bursaphelenchus xylophilus (Steiner&Buhrer) Nickle. In: Zhang XY, Luo YQ (Eds) Major Forest Diseases and Insect Pests in China.Beijing: China Forestry Publishing House, 1–29.
- Yin M, Wingfield MJ, Zhou X, Beer ZWD (2016) Multigene phylogenies and morphological characterization of five new Ophiostoma spp. associated with Spruce-infesting bark beetles in China. Fungal Biol 120(4): 454–470. https://doi.org/10.1007/s11557-009-0594-4
- Zhao L, Sun J (2017) Pinewood Nematode Bursaphelenchus xylophilus (Steiner and Buhrer) Nickle//Biological Invasions and Its Management in China. Springer, Singapore, 3–21. https://doi.org/10.1007/978-981-10-3427-5_1
- Zhao L, Mota M, Vieira P, Butcher RA, Sun J (2014) Interspecific communication between pinewood nematode, its insect vector, and associated microbes. Trends in parasitology 30(6): 299–308. https://doi.org/10.1016/j.pt.2014.04.007
- Zhao L, Lu M, Niu H, Fang G, Zhang S, Sun J (2013) A native fungal symbiont facilitates the prevalence and development of an invasive pathogen-native vector symbiosis. Ecology 94(12): 2817–2826. https://doi.org/10.1890/12-2229.1
- Zhao BG, Futai K, Sutherland JR, Takeuchi Y (2008) Pine Wilt Disease. Springer, Tokyo, 144–161. https://doi.org/10.1007/978-4-431-75655-2
- Zhao GH, Chen XY, Wu YZ (2006) Study on the Fungi from the Diseased Pine Wood Nematode. Journal of Nanjing Forestry University (Natural Sciences Edition) 30(2): 79–81.
- Zhao GH (1992) Two new species of the genus Ceratocystis. Journal of Nanjing Forestry University 16(2): 81–86.
- Zhou XD, Burgess T, de Beer ZW, Wingfield BD, Wingfield MJ (2002) Development of polymorphic microsatellite markers for the tree pathogen and sapstain agent, Ophiostoma ips. Molecular Ecology Resources 2(3): 309–312. https://doi.org/10.1046/j.1471-8286.2002.00225.x-i2
- Zhou X, Burgess TI, de Beer ZW, Lieutier F, Yart A, Klepzig K, Carnegie A, Portales JM, Wingfield BD, Wingfield MJ (2007) High intercontinental migration rates and population admixture in the sapstain fungus Ophiostoma ips. Molecular Ecology 16(1): 89–99. https://doi.org/10.1111/j.1365-294X.2006.03127.x
- Zhou X, de Beer ZW, Wingfield MJ (2006) DNA sequence comparisons of Ophiostoma spp., including Ophiostoma aurorae sp. nov., associated with pine bark beetles in South Africa. Stud Mycol 55: 269–277. https://doi.org/10.3114/sim.55.1.269
- Zhou X, de Beer Z, Cibrian D, Wingfield BD, Wingfield MJ (2004) Characterisation of Ophiostoma species associated with pine bark beetles from Mexico, including O. pulvinisporum sp. nov. Mycological Research 108(Pt 6): 690–698. https://doi.org/10.1017/S0953756204009918
- Zipfel RD, Beer ZW de, Jacobs K, Wingfield BD, Wingfield MJ (2006) Multigene phylogenies define Ceratocystiopsis and Grosmannia distinct from Ophiostoma. Studies in Mycology 55: 75–97. https://doi.org/10.3114/sim.55.1.75
Supplementary materials
Figure S1. Phylogram of fungal associates of pine infected by PWN and Monochamus alternatus in China
Figure S2. Phylograms of fungal associates of pine infected by PWN and Monochamus alternatus in China
Figure S3. Three ML phylogenetic threes based on tub2 after excluding introns