- ContentsContents
- Article InfoArticle Info
- CiteCite
- MetricsMetrics
- CommentComment
- RelatedRelated
- FigsFigs
- TaxaTaxa
- RefsRefs
- CitedCited
- NanopubsNanopubs
Bipartite and tripartite cyanolichens. A In the bipartite cyanolichen Peltigera scabrosa the cyanobacterial symbiont (Nostoc) forms a continuous layer just below the upper cortex of the lichen thallus B Nephroma bellum is another example of bipartite cyanolichens C In the tripartite cyanolichen Peltigera aphthosa the Nostoc symbiont is restricted to wart-like cephalodia (shown magnified) on the upper surface of the thallus, while the green algal symbiont (Coccomyxa) forms the photobiont layer D Nephroma arcticum is another example of tripartite cyanolichens. The large cephalodia of this species are internal, but clearly visible through the upper cortex of the hydrated thallus.
Free-living and lichen-symbiotic Nostoc strains. A Free-living Nostoc colonies and gelatinous cyanolichens (Collema sp.) growing on mineral soil in northern Spain B Free-living Nostoc colony on limestone in northern Italy C Lichenized Collema thallus (containing Nostoc symbionts) on limestone in northern Italy D–F Morphological variation of Nostoc symbionts inside two Leptogium thalli in southwestern Kenya. The large clear cells are nitrogen-fixing heterocysts, the smaller translucent structures are fungal hyphae in optical cross-section.
Examples of cyanolichens examined in molecular studies of cyanobacterial diversity. A Cephalodial symbionts of the tripartite cyanolichen Lobaria pulmonaria remain poorly known (e.g. Rikkinen et al. 2002, Myllys et al. 2007) B Nostoc symbionts of Pseudocyphellaria species have been analyzed in several studies (e.g. Summefield et al. 2002, 2006, Rikkinen et al. 2002, Stenroos et al. 2006) C Peltigera venosa may have different Nostoc genotypes in different cephalodia (Paulsrud et al. 2000) D Nostoc symbionts of bipartite Peltigera species have been identified in many studies (e.g. Paulsrud and Lindblad 1998, O’Brien et al. 2005, Kaasalainen et al. 2012) E Otalora et al. (2010) analyzed genetic diversity of Nostoc in Collema and related cyanolichens F Many tropical Leptogium specimens were screened by Kaasalainen et al. (2012) G A Nostoc strain isolated from Pannaria pezizoides produces potent hepatotoxins in culture (Oksanen et al. 2004, Kaasalainen et al. 2009) H The cyanobacterial symbionts of Coccocarpia species are only distantly related to Nostoc (Lücking et al. 2009).
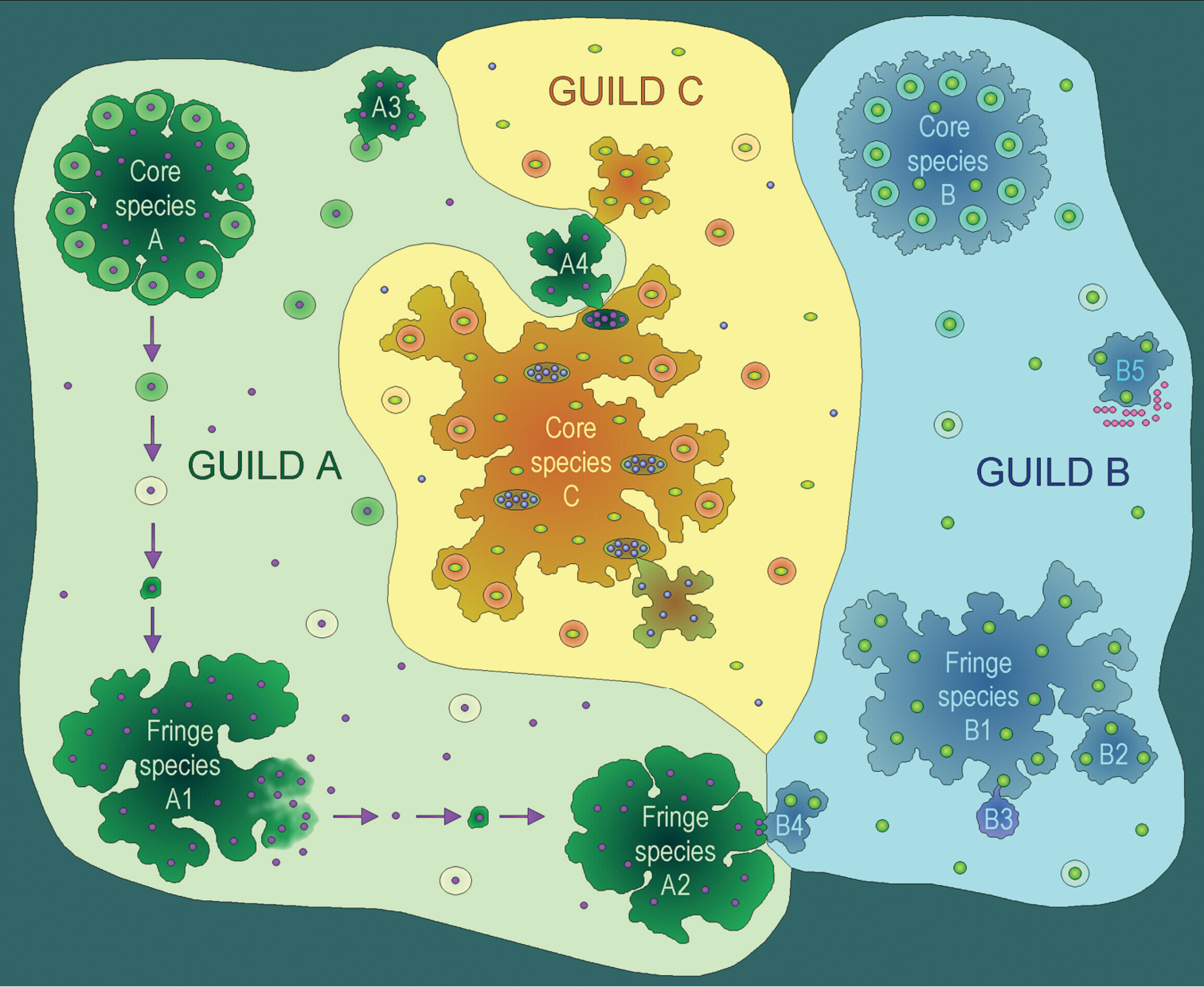
Photobiont-mediated guilds in lichens (modified from Rikkinen 2003).The lichen-forming fungi belong to three different guilds, one centring around cyanobacterial symbionts (A), the second around green algal symbionts (B), and the third around another genus of green algal symbionts (C). The lichen in the middle of the picture houses both green algae and cyanobacteria (in cephalodia), meaning that its fungal symbiont can operate in two different guilds (C and A). As the symbiotic propagules of this lichen only contain the fungus and green algal photobiont, the fungus is a core species in guild C and a fringe species in guild A. Under certain conditions this fungus may give rise to cyanobacterial morphotypes (A4) and/or green algal thallus lobes. The core species of the lichen guilds produce innumerable symbiotic propagules, most of which will never develop into mature thalli of that lichen species. Germinating spores of fringe species (A1–A4, B1–B5) may commonly acquire their photobionts from small free-living populations that originate from disintegrating symbiotic propagules of the core species. At the latest when the thallus of a fringe species dies and disintegrates (A1), some of the photobionts are released back to the local environment for the common benefit of all fungi of the same guild. However, without the ability to produce symbiotic propagules, the fringe species cannot effectively disperse appropriate photobionts into new habitats. Some fringe species are aggressive enough to steal photobionts from juvenile stages or weakened thalli of other lichen species (A3), or live as lichenicolous lichens (B2) on other lichens of the same guild. The juvenile stages of some green algal lichens establish loose cyanotrophic associations with free-living cyanobacteria (B5) and/or cyanolichens (B4). Some lichenicolous fungi (B3) have evolved from lichen-forming ancestors and in many cases also their host ranges still appreciate guild boundaries.
Environments sampled in molecular studies of cyanobacterial diversity. A Arctic tundra on Svalbard; so far only two studies have included cyanolichen specimens from polar environments (Wirth et al. 2003, Kaasalainen et al. 2012) B Boreal forest in central Finland; cyanolichens from boreal forests have been examined in several studies (e.g. Paulsrud and Lindblad 1998, Myllys et al. 2007, Fedrowitz et al. 2011) C Temperate forest in western North America; also cyanolichens from temperate forests have been analyzed in several studies (e.g. Rikkinen et al. 2002, Summerfield et al. 2002, Fedrowitz et al. 2011) D Tropical montane forest in East Africa; so far only two studies have included cyanolichen specimens from tropical ecosystems (Lücking et al. 2009, Kaasalainen et al. 2012).
Plant hosts of symbiotic cyanobacteria; some of which can share Nostoc symbionts with cyanolichens. A The liverwort Blasia pusilla has Nostoc symbionts in auricles (Rikkinen and Virtanen 2008) B Hornworts house Nostoc symbionts in slime cavities (Costa et al. 2002) C The cyanobacterial symbiont of the water fern Azolla is not closely related to lichen symbiotic cyanobacteria (Ran et al. 2010) D All cycads associate with cyanobacteria, mainly Nostoc (Costa et al. 1999) E The cyanobacterial symbionts of cycads are housedin specialized roots (Costa et al. 2004, Gehringer et al. 2010, Yamada et al. 2012) F Gunnera species have endosymbiotic Nostoc in creeping rhizomes (Nilsson et al. 2000).